That type of atom is called an isotope. Every chemical element has one or more isotopes. An atom is first identified . There are 2isotopes of the stable elements, in addition to over 8radioactive isotopes.
Not all atoms of an element are identical – atoms of the same element can have different numbers of neutrons.

These different versions of the same element are called isotopes. In this lesson, we will discuss the examples and types of isotopes. With this option, your source code is kept proprietary. Are all atoms of an element the same? How can you tell one isotope from another ? Use the sim to learn about isotopes and how abundance relates to the average atomic mass of an element.
This is the definition of an isotope along with examples. The different isotopes of an element have identical chemical properties.
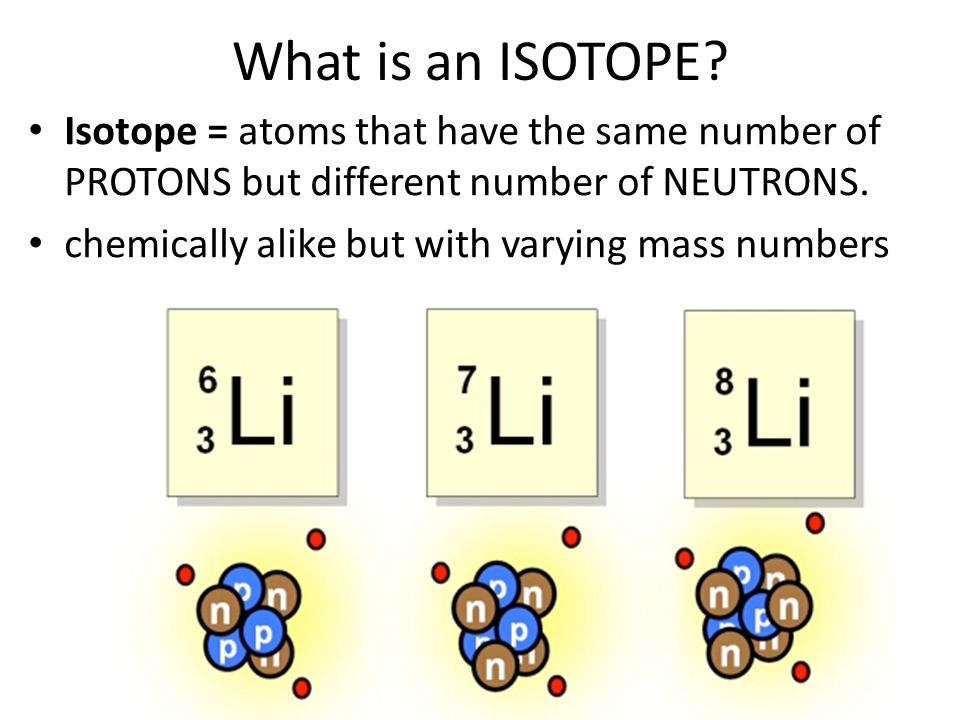
However, some isotopes are radioactive. You can think of isotopes as atomic siblings. They are part of the same family (same number of PROTONS) but they are unique in that each isotope has a different number of NEUTRONS in its nucleus. The MASS NUMBER of any ion is the sum of its PROTONS and NEUTRONS. Atoms that have the same number of protons but different numbers of neutrons are called isotopes.
Isotopes – Higher tier. The element hydrogen, for example, has three commonly known isotopes : protium, deuterium and tritium. This page is maintained by Steve Gagnon.
In this educational animated movie about Science learn about elements, neutrons, uranium-23 isotopes , carbon-1 and atoms. To understand nuclear issues, you must have a basic understanding of what exactly an isotope is. An isotope is one particular form an atom of an element might take. Just as people may be thin, medium weight, or heavy, isotopes vary according to their weight.
You will remember from the last chapter that the mass of an atom is determined by adding the number of protons to the number of neutrons in . Visit us to learn more. Hanging out, sketching, signing, and having a great Saturday afternoon with us. These two artists represent everything we think is exciting about the future of comics. Stop on by and show these amazing creators some well- deserved love .

Nov Physicists show that thunderstorms trigger nuclear reactions in the atmosphere. Categories: English non-lemma forms . Two or more forms (or atomic configurations) of a given element that have identical atomic numbers (the same number of protons in their nuclei) and the same or very similar chemical properties but different atomic masses ( different numbers of neutrons in their nuclei) and distinct physical properties. Common elements that possess isotopes include carbon, oxygen, hydrogen, and nitrogen.